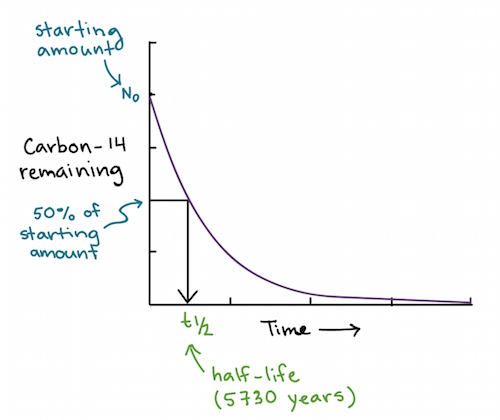
A radiaoactive nucleus (initial mass number `A` and atomic number `Z` emits `3 a- particles - YouTube

How To Calculate Relative Atomic Mass - What is Relative Mass? How to Calculate Relative Atomic Mass along with FAQs

Atomic Ratios in Compounds | How to Find the Number of Atoms in a Compound - Video & Lesson Transcript | Study.com
Modern Periodic Table of the Elements with atomic number, element name, element symbol and atomic mass, in english language Stock Vector | Adobe Stock

A radioactive nucleus (initial mass number A and atomic number Z) emits 3 alpha - particles and 2 positrons. The ratio of number of neutrons to that of protons in the final

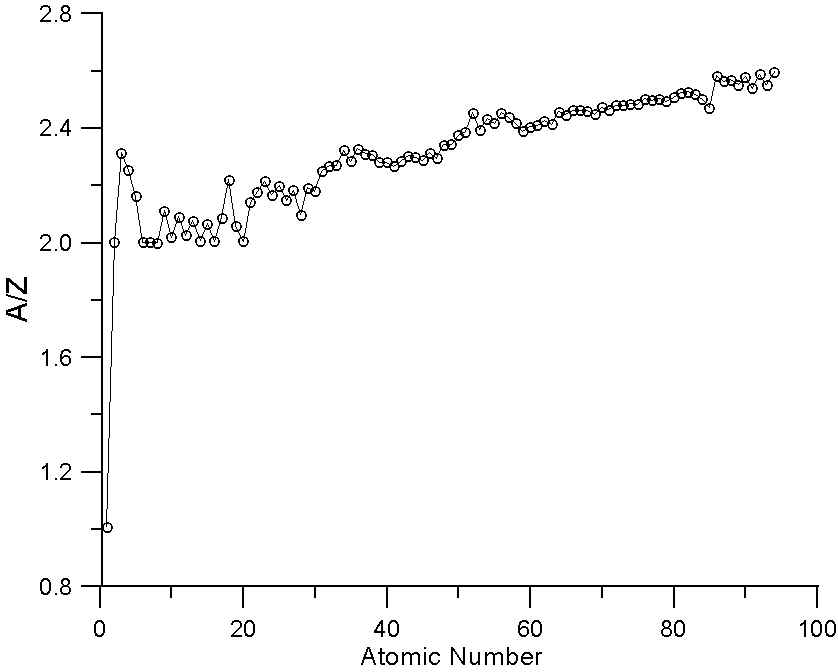
Color) Ratio R E 1 ; E 2 ; t; Z† vs atomic number Z for barriers with... | Download Scientific Diagram

The atomic mass of lead is 208 and its atomic number is 82. The atomic mass of bismuth is 209 and its atomic number is 83. The ratio of neutrons/protons in the atom






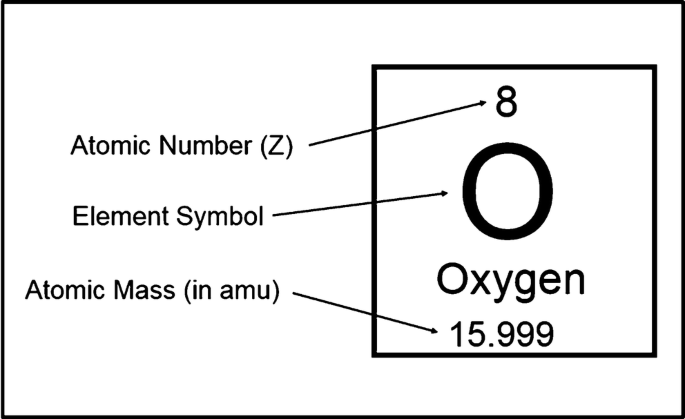



:max_bytes(150000):strip_icc()/boron-illustration-545864379-5838819f5f9b58d5b1c57b5f.jpg)